Ritesh Indulkar / August 28, 2025 / Pharmaceuticals Blogs
Ethiopia’s Step Towards Pharmaceutical Track & Trace Compliance
As of August 19, 2025, Ethiopia is well on its way to transforming its pharmaceutical supply chain with the launch of its national traceability system.
The Ethiopian Food and Drug Authority (EFDA) has recently rolled out a series of announcements that are setting the stage for stricter compliance, greater visibility, and enhanced patient safety.
With deadlines now approaching fast, stakeholders—particularly exporters—must act quickly to align with the EFDA’s new requirements.
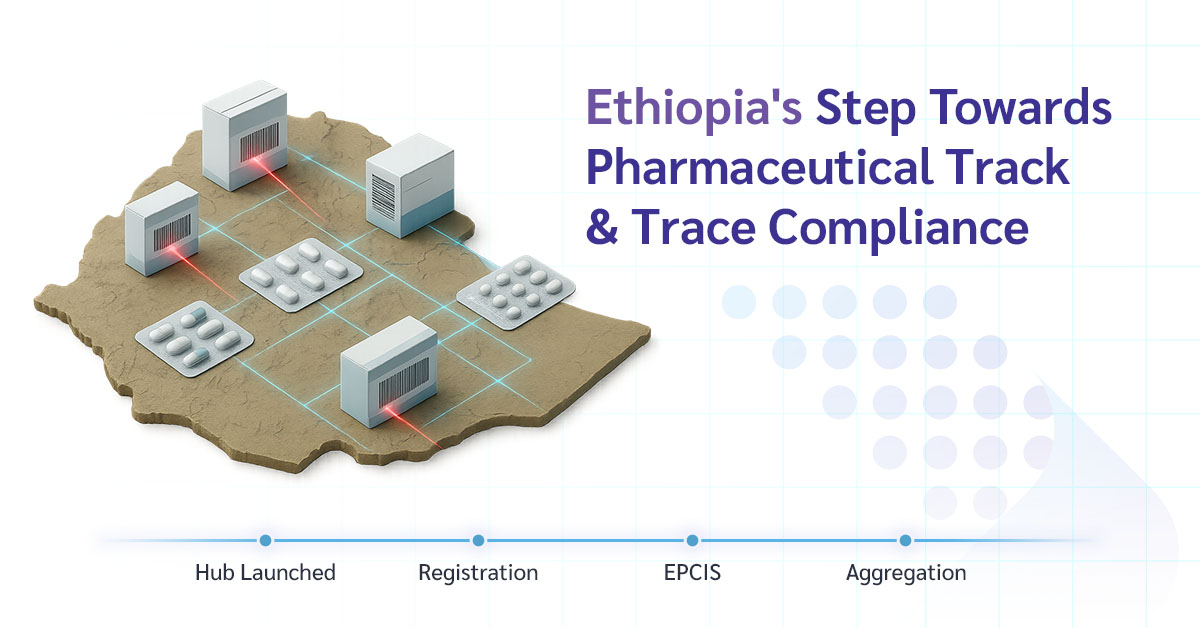
Key Updates from EFDA
1. Traceability Hub Live Since June 26, 2025
The EFDA’s centralized Traceability Hub officially went live earlier this summer.
This platform is now the backbone of Ethiopia’s serialization and reporting framework, and all supply chain stakeholders must connect to it to exchange compliance data.
2. Registration Deadline Looming
Pharmaceutical manufacturers and exporters to Ethiopia have until July 25, 2025, to register on the EFDA portal.
That deadline has already passed, so any company not yet registered is now at risk of non-compliance and potential disruption of market access.
3. Mandatory EPCIS Event Reporting Coming Up
The next major milestone is June 3, 2026, when all exporters will need to start reporting serialization and supply chain events using EPCIS standards.
This covers commissioning, shipping, receiving, and decommissioning serialized packs.
4. Aggregation Deadline on the Horizon
Aggregation reporting is currently optional, but it will become mandatory by December 1, 2026.
That gives companies just over a year to prepare parent-child packaging visibility between units, cartons, and pallets.
Why This Matters Right Now
The EFDA’s timeline shows that Ethiopia is serious about aligning with global best practices in pharmaceutical traceability.
For companies in the supply chain, these rules are about much more than compliance:
- Patients gain protection from counterfeit and unsafe products.
- Authorities gain oversight across every stage of distribution.
- Companies gain trust and efficiency by digitizing their supply chain processes.
The takeaway? If you’re not already registered and planning your EPCIS and aggregation readiness, the clock is ticking.
Compliance Timeline at a Glance
| Date | Requirement / Milestone |
| June 26, 2025 | EFDA Traceability Hub launched |
| July 25, 2025 | Registration deadline for exporters (already passed) |
| June 3, 2026 | Mandatory EPCIS event reporting begins |
| December 1, 2026 | Aggregation event reporting becomes mandatory |
How Jekson Can Support
At Jekson, we understand that compliance is only part of the journey—companies also need reliable partners who can deliver end-to-end solutions.
What makes Jekson unique is that we are one of the very few companies globally to develop and deliver both hardware and software solutions internally, covering the full track and trace ecosystem from Level 1 (line devices) to Level 5 (enterprise systems).
- Hardware Expertise (Level 1 & 2): Jekson designs and manufactures high-precision serialization and aggregation equipment, ensuring packaging lines are compliance-ready.
- Software Integration (Level 3 to 5): From site-level serialization management to enterprise-wide reporting and regulatory compliance, our in-house software platforms ensure seamless connectivity, EPCIS reporting, and data integrity.
- End-to-End Ownership: Because everything is built under one roof, our clients benefit from unified support, faster implementation, and solutions that are fully harmonized across every compliance layer.
This holistic approach makes Jekson a trusted partner for companies preparing for Ethiopia’s compliance journey and beyond.
The Way Forward for the Industry
With today’s date—August 19, 2025—companies are already in the critical implementation window.
Here’s what needs to happen next:
- Check registration status: Any exporter not yet onboarded must urgently resolve this to avoid supply chain disruptions.
- Start EPCIS integration: Waiting until 2026 is risky. Building EPCIS reporting capability now ensures smoother transitions.
- Plan aggregation systems: Companies should begin investing and piloting aggregation solutions well before the mandatory deadline.
At Jekson, we view Ethiopia’s regulations not only as a compliance requirement but also as an opportunity to embrace digital supply chains.
With our combined hardware and software capabilities, we provide a single-window solution that ensures companies are not just compliant but also future-ready.
Final Thought
The message is clear: Ethiopia is moving fast, and the pharmaceutical industry must keep pace.
As of August 2025, the foundation has already been laid.
Now it’s about execution. Companies that take proactive steps today—supported by partners like Jekson—will be ready for tomorrow’s fully digital, transparent, and secure pharmaceutical landscape.
To learn more or explore collaboration opportunities, contact us at marketing@jeksonvision.com or call/WhatsApp us on +91 9328233954







